
Liver-preferential insulin analogue
Category
Insulin
ID
NNC0123-0327
Amount per vial
1300 nmol
Insulin is a peptide hormone acting as a key regulator of glucose homeostasis. Insulin binds to the insulin receptor (InsR), which exists in two alternatively spliced isoforms, InsR-A and InsR-B. The insulin receptor belongs to the same family of receptor tyrosine kinases as the IGF1 receptor (IGF1R) and insulin is therefore also able to bind to the IGF1R albeit with considerably lower affinity as compared to the insulin receptor.
NNC0123-0327 is a liver-preferential insulin analogue with a C20 fatty diacid attached at position A22K. NNC0123-0327 is able to bind reversibly to serum albumin with high affinity (which reduces the trans-endothelial transport from plasma to periphery) while retaining a high InsR affinity in the presence of albumin compared to other acylated insulin analogues ensuring a high hepatic InsR-mediated clearance). Due to these properties, NN0123-0327 therefore displays its liver-preferential effects following peripheral administration.
Category
Insulin
ID
NNC0123-0327
Amount per vial
1300 nmol
| Property | NNC0123-0327 | Human insulin |
| MW (Da) | 6607 | 5808 |
| pI | 4.8 | 5.7 |
| Fatty acid | C20 diacid | - |
| Linker | gGlu-2xOEG | - |
| Sequence substitutions | A22K, B29R, desB30 | - |
| Sum Formula | C296 H453 N71 O88 S6 | C257 H383 N65 O77 S6 |
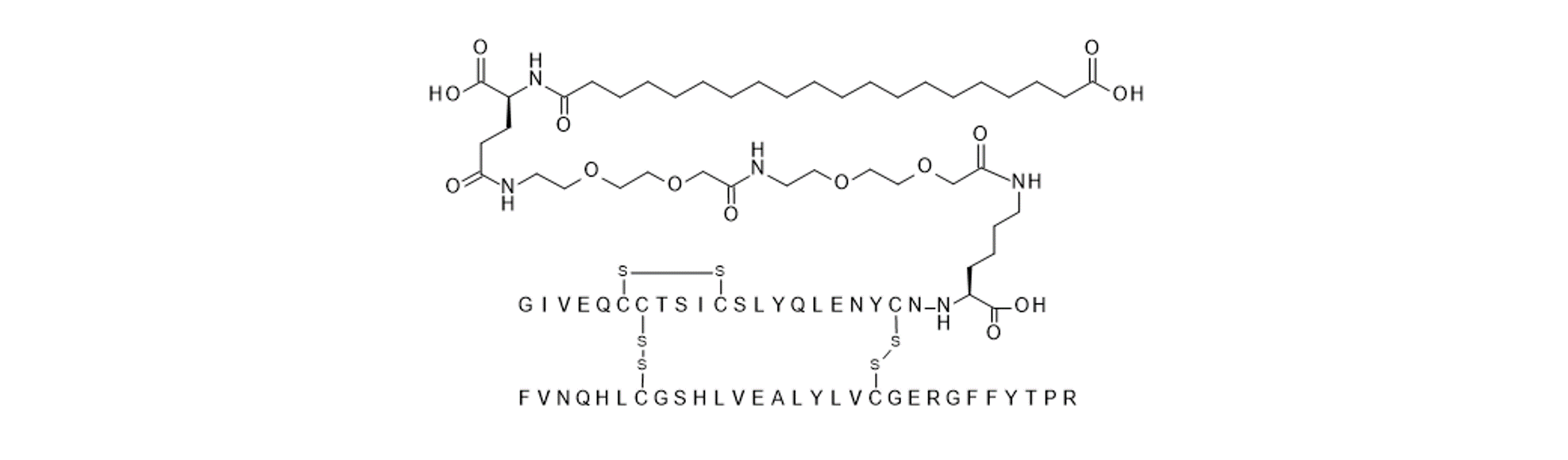
Figure 1
2D sketch of NNC0123-0327. The B chain is lacking threonine at position B30, contains a B29 arginine substitution and there is an additional lysine a position A22 to which a fatty diacid is attached via a gamma-Glu-2xOEG linker.
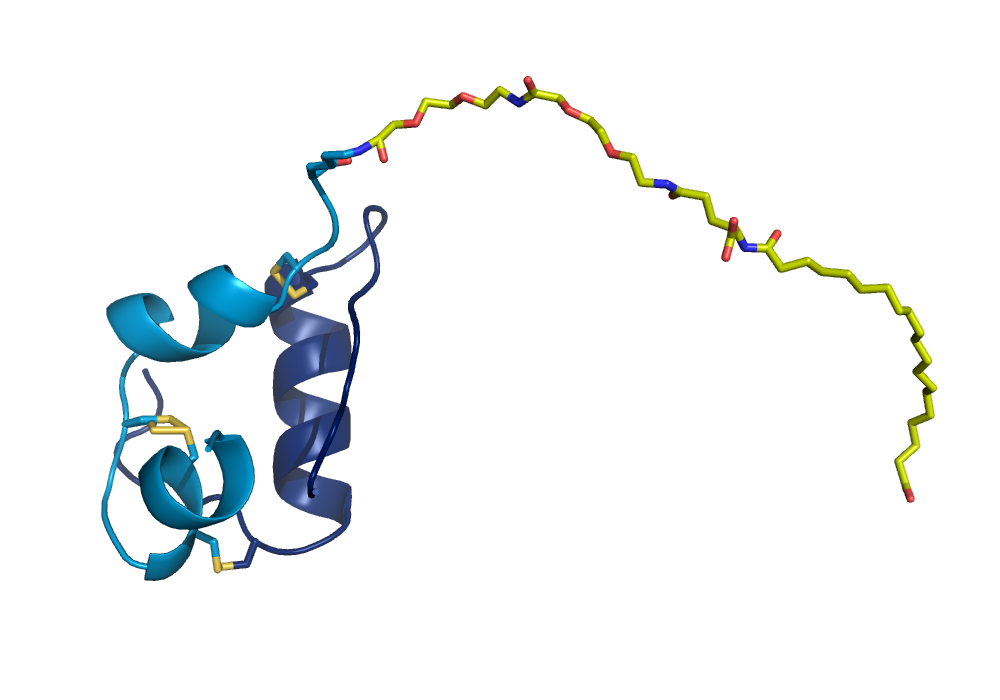
Figure 2
Graphic representation of the crystal structure of NNC0123-0327 crystallised under zinc free conditions (unpublished structure). The fatty acid, the linker and the lysine at position A22K are shown in yellow stick representations.
The in vitro receptor binding affinity of NNC0123-0327 relative to human insulin (HI) is shown in the table below. The affinity was determined using human insulin receptor isoform A and also the human IGF1R as solubilized holoreceptors in the absence or presence of 1.5% (w/v) human serum albumin (HSA). Since albumin binding is a key mechanism for the design of NNC0123-0327, be aware that the apparent affinity and potency will be very dependent on whether the in vitro assays contain albumin or not.
| Receptor | HSA concentration [%] | Relative affinity (mean +/- SD) [% of HI] |
| Human InsR-A | No HSA | 165 +/- 26.5 |
| 1.5 | 11.3 +/- 1.1 | |
| Human IGF1R | No HSA | 570.3 +/- 47.4 |
| 1.5 | 9.1 +/- 0.1 |
The pharmacokinetic properties of NNC0123-0327 in Wistar rats following an i.v. injection are listed in the table.
NNC0123-0327 displays a markedly protracted PK profile in vivo following a s.c. injection. Plasma PK parameters following i.v. dosing from a study in Wistar rats are given in the table below. For i.v. and s.c. studies in rats, dose levels of 1-5 nmol/kg and 5-30 nmol/kg, respectively, are recommended both for once and twice daily dosing studies. For the i.v. study in the table below, NNC0123-0327 was dissolved in 5 mM phosphate, 140 mM NaCl and 70 ppm polysorbate 20, pH 7.4. The PK parameters for NNC0123-0327 are determined in wake rats whereas the parameters for human insulin are determined in anaesthetized rats. The evaluation of the liver-preferential effects of NNC0123-0327 is described in the Edgerton et al. references listed in the reference section.
| Parameter | NNC0123-0327 | Human insulin |
| Dose [nmol/kg)] | 5 | 1.2 |
| T1/2 [h] | 0.83 | 0.28 |
| MRT [h] | 0.68 | 0.15 |
| CL [mL/min/kg] | 0.96 | 22 |
| Vz (mL/kg) | 78 | 623 |